August 7, 2018 – Researchers at Dartmouth College have revealed how a key protein functions during the millions of cell divisions that occur in the human body each minute. The research describes two separate but coordinated pulling actions generated by the protein dynein that ensure healthy cell division in humans and other organisms.
| Image 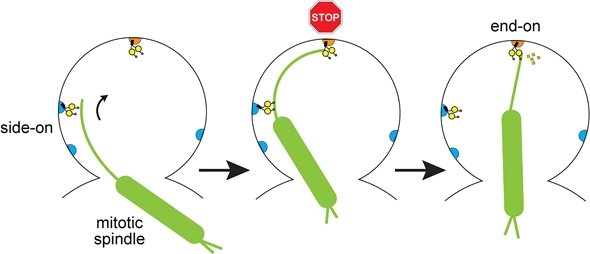 |
| Dynein (shown in yellow) applies “side-on” and “end-on” pulling forces to position the mitotic spindle during cell division in baker’s yeast. Illustration courtesy of Wei-Lih Lee. |
The Dartmouth research demonstrates for the first time that dynein employs both “side-on” and “end-on” forces to carry out its work during mitosis. The study also shows that dynein chooses which mechanism to activate depending on where the protein is located within the cell.
The study, published in the journal eLIFE, focuses on dynein in baker’s yeast, an organism that features cells that function similarly to those in the human body. The research adds to the understanding of the mechanisms involved in cell reproduction and sheds light on a cellular-level mystery that has confounded researchers.
“For close to two decades researchers have strained to understand why dynein appears to behave differently in yeast,” said Wei-Lih Lee, a professor of biological sciences at Dartmouth. “The discovery of the protein’s dual role answers this question and allows us to learn even more about the regulation of dynein during cell division.”
Dynein – referred to as an “ancient motor protein”– is widely known for its role in carrying waste products from nerve terminals back to cell bodies in the spinal cord. The protein is also essential for processes like embryonic development and the steady maintenance of stem cells in the basal layers of the skin.
During cell division, dynein positions the mitotic spindle, a complex apparatus that allows cells to segregate genetic material. Once the spindle is aligned by dynein, DNA is distributed equally and the two daughter cells can survive.
“Positioning the mitotic spindle correctly is absolutely essential in order to have healthy cells, and it’s dynein that performs that task,” said Safia Omer, a graduate research assistant who was the lead author of the study.
To do its work, dynein uses two distinct pulling mechanisms. The first of these mechanisms, “side-on” pulling, applies lateral force to the mitotic spindle, similar to pulling on the side of a rope. The second mechanism, “end-on” pulling, pulls the spindle from its end, similar to the way a wood chipper draws in a log. This end-on pulling action also applies a brake to the spindle’s movement and causes it to set in precisely the correct position.
Until the Dartmouth study, researchers were uncertain as to why dynein seemed to prefer one mechanism over the other depending on which organism it was in.
“We were puzzled as to why the ‘end-on’ pulling action didn’t appear to exist in yeast, but it actually does. It was evading discovery for all of this time because this mechanism only exists for a short time and in a small location in the cell,” said Lee.
In addition to discovering this dual-role for dynein, the Dartmouth research demonstrates that the protein only activates “side-on” pulling when it sits on the sides of the cell membrane, and pulls from the end when it sits on the apex of the cell. The study also uncovers an unexpected role for the cellular structure known as the endoplasmic reticulum in regulating dynein behavior.
According to the study, the role of dynein is most essential when creating asymmetric daughter cells, with one transforming into a tissue cell and the other remaining as a stem cell. If a mutation causes dynein to position the spindle incorrectly, the daughter cells would feature an abnormal fate leading to maladies like skin cancer or the “smooth brain” disease lissencephaly.
“Basic science such as this is an investment in the future,” said Samuel Greenberg, an undergraduate student at Dartmouth who assisted with the research. “Understanding these processes could allow researchers to make candidate drugs to target specific problems in the pathway.”
Future research will focus on how dynein knows where to attach itself within the cell plasma to deploy the required pulling forces for the critical process of cell division.
For additional information, please contact Wei-Lih Lee at: wei.lih.lee@dartmouth.edu

